During the last three decades, our work has focused on and contributed to the area of protein and membrane trafficking through the molecular analysis of the pathogenesis of intestinal diseases and has unraveled molecular mechanisms implicated in these processes. By utilizing tissues from patients with intestinal carbohydrate malabsorption disorders, intestinal Caco-2 or HT-29 cells, or stably transfected polarized or non-polarized cells, a large body of knowledge in the molecular basis of intracellular trafficking and protein folding mechanisms has accumulated. Most important discoveries are related to the following lines of research:
- Cellular mechanisms in intestinal cell biology relevant to polarized and vesicular trafficking of intestinal proteins, implication of N- and O-linked glycosylation and lipid microdomains in the intracellular targeting of intestinal proteins and quality control mechanisms;
- Genetic, cellular and biochemical basis of the intestinal disorders, congenital sucrase-isomaltase deficiency (CSID) and congenital lactase deficiency (CLD) and definition of these diseases as protein trafficking and folding diseases; establishment of the concept of symptoms heterogeneity and the existence of a gradient of severity in CSID;
- Functional variants of sucrase-isomaltase potentially implicated in irritable bowel syndrome (IBS);
- Cellular and biochemical mechanisms in the pathogenesis of microvillus inclusion disease (MVID);
- Targeting and endocytic mechanisms of gliadin peptides in the pathogenesis of Celiac disease.
Current Projects
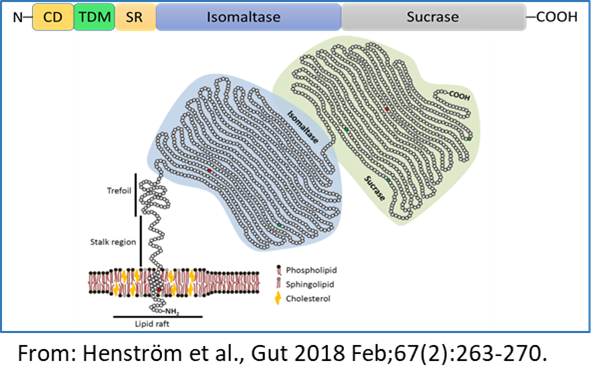
The multi-faceted genetically-determined carbohydrate malabsorption in congenital sucrase-isomaltase deficiency and irritable bowel syndrome
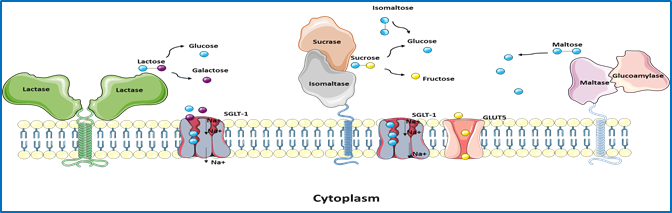
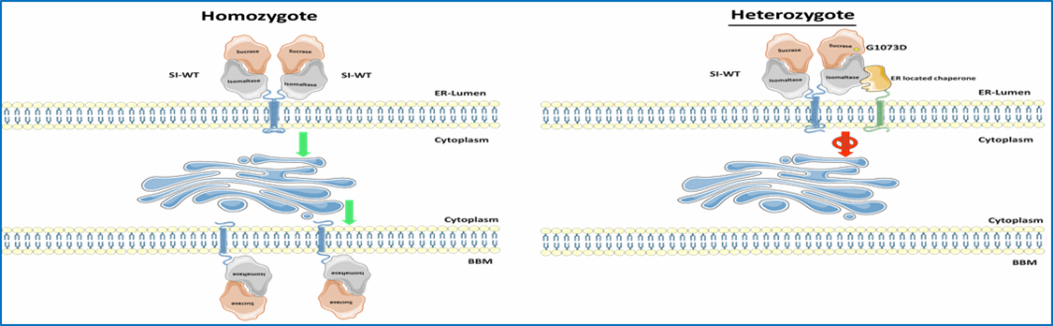
The final stages of the digestion of α-glycosidic carbohydrates occurs in the small intestine through the α-glycosidases sucrase-isomaltase (SI, EC 3.2.148 and 3.2.1.10) and maltase-glucoamylase (MGAM¸ EC 3.2.1.20 and 3.2.1.3). Reduced expression levels or complete absence of intestinal disaccharidases at the cell surface of the enterocytes is associated with carbohydrate maldigestion and malabsorption, most notably described in several cases of genetically-determined sucrase-isomaltase deficiency (CSID). Unlike CSID, irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder (FGID) that affects 10-15% of adults and children worldwide. Biochemical, cellular and functional analyses of SI mutations established the concept of phenotypic heterogeneity of CSID and classified SI mutants based on their intracellular location and function as well as the genetic inheritance pattern, homozygous or combined heterozygous. The concept of heterozygosity in CSID has been also recently discussed.
Due to several common symptoms with CSID, such as diarrhea, abdominal cramping and flatulence, a potential role of SI (and probably also MGAM) in the pathogenesis of IBS-D (D = diarrhea) has been proposed.
In this project we want to investigate the role of SI and MGAM in the pathogenesis of genetically determined carbohydrate malabsorption in CSID and IBS.
- We will first analyze the protein targeting and activity profiles of SI mutants in heterozygous inheritance pattern in CSID in comparison to wild-type SI. In parallel studies the influence of MGAM on the overall carbohydrate digestive capacity in this cellular model will be assessed.
- In a similar experimental set up we will study SI polymorphisms in IBS and their potential association with MGAM. For this purpose, the widespread gene variant V15F and other pathogenic SI variants with reduced enzymatic activities in IBS patients will be investigated.
- A wide screen will be envisaged to identify polymorphisms of SI and other carbohydrate digesting enzymes (CDE), (e.g. MGAM, lactase, pancreatic amylase) in patient samples and controls from various EU and US neurogastroenterology centers using targeted next generation sequencing. Following similar procedures as above, the effects of potential mutations found in these enzymes on their intracellular targeting and enzyme function will be analyzed with the ultimate goal of obtaining an unequivocal basic knowledge of the etiology of IBS relevant to CDE.
These studies are expected to fundamentally expand our understanding of the molecular basis of carbohydrate malabsorption in functional gastrointestinal disorders.
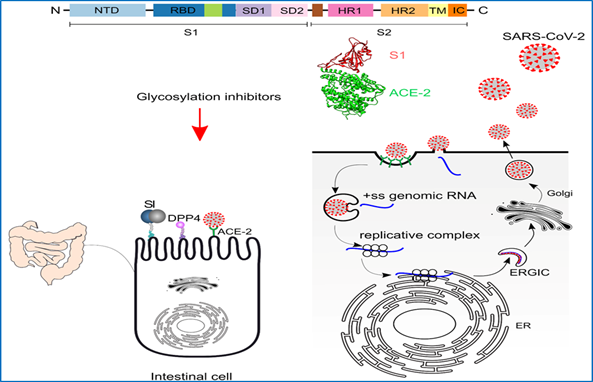
Infection of intestinal cells by SARS-CoV2 and its effects on intestinal function
CoVid-19 is characterized by infection of the airways by SARS-CoV-2. Apart from the respiratory tract, other organs are also involved, e.g. the intestinal tract. The importance of the intestinal infection is increasingly recognized. In a large proportion of pediatric patients, virus was detected in rectal swabs and virus shedding from the intestine was found even when oral swabs had become negative. Therefore, prolonged virus shedding and fecal-oral transmission have to be considered. This notion is supported by detection of the virus in wastewater.
The aim of this project is to apply intestinal cell cultures to characterize the infection of differentiated intestinal epithelial cells by SARS-CoV-2 and thereafter intestinal orgranoids.
This in vitro infection approach will target the following aims:
1. Characterization of the replication efficiency of SARS-CoV-2 (virus yield, virus exit, virus entry, apical, basolateral).
2. Localization of the cellular receptor(s) in human intestinal Caco-2 cells
3. Investigation of the trafficking of the cellular receptor(s), determination and subsequent modulation of their mode of interaction with membrane microdomains (lipid rafts, LRs)
4. Effects of glycosylation modulators on the spike glycoprotein and its interaction with intestinal cells
5. Implication of virus infection on the trafficking and function of crucial enzymes of the intestinal physiology (APN, SI, LPH, DPP4).
This project will provide substantial information on the replication of SARS-CoV-2 in intestinal epithelial cells, evaluate its effects on the intestinal function and provide solid hypotheses on the molecular and biochemical basis for the symptoms elicited by SARS-CoV-2 infections. These hypotheses can be then examined at a later stage in intestinal organoids. Further, unravelling the biosynthetic pathway, glycosylation pattern and mode of interaction of the SARS-CoV-2 receptors and its modulation could constitute exquisite targets for potential therapy.
Biochemical and cellular basis of carbohydrate malabsorption in microvillus inclusion disease and potential rescue of the brush border membrane function
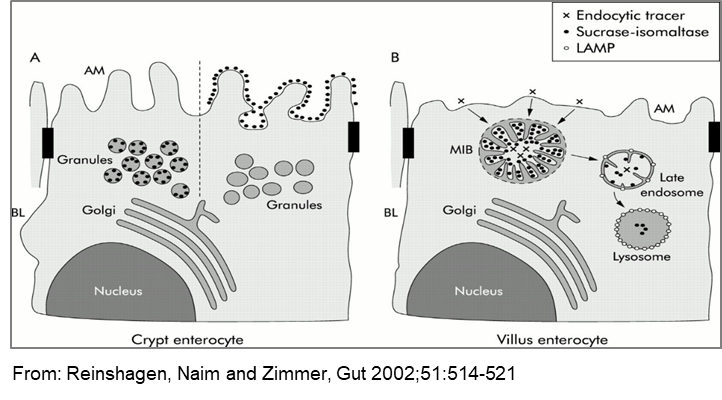
Microvillus inclusion disease (MVID) is a rare congenital disorder that is characterized by a villus atrophy and the presence of microvillus inclusion bodies (MVIBs). In experimental settings using cationized ferritin in human biopsies we have previously demonstrated that the inclusion bodies originate from autophagocytosis of apical cell membrane and represent early endosomes. The genetic defect in MVID has been meanwhile mapped to the MYO5B gene that encodes the unconventional type myosin 5b motor protein. Myosin 5b is associated with the plasma membrane recycling system in non-polarized cells and the apical recycling system in polarized cells. Furthermore, it has been shown that the Rab11 family interacting protein 2 associates with myosin 5b in regulating plasma membrane recycling. These findings underline the importance of unravelling the cellular mechanisms of autophagocytosis of the apical membrane of enterocytes in MVID membranes with the ultimate goal of designing appropriate therapeutic measures in this context. In this proposal we will generate intestinal Caco-2 cells in which myosin 5b has been downregulated, depleted or modified and assess at the molecular and biochemical levels the consequences on the trafficking and targeting of a panel of brush border membrane proteins.
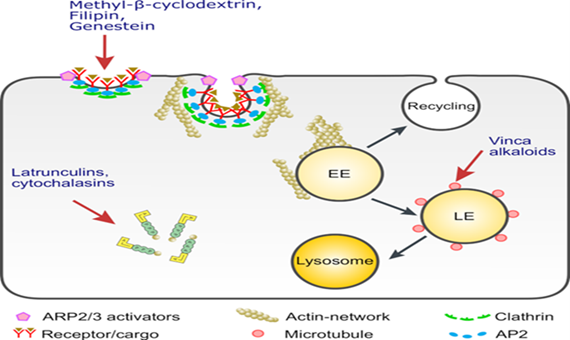
We will further investigate the function of inhibitors of protein transport and polarized sorting as well as vesicular trafficking in myosin 5b-depleted Caco-2 cells as potentiators of brush border membrane rescue. The data gained from the cell culture model will be confirmed and expanded into intestinal organoids that will be generated from intestinal biopsy specimens of MVID patients. In this model we will address similar questions to those in the cellular model and identify targets for (i) preventing the formation of MVIBs and (ii) potential restoration of proper trafficking and sorting of intestinal protein markers and reducing carbohydrate malabsorption. The ultimate goal is to test inhibitors and modulators of these targets that are the least toxic for enterocytes. These substances would then represent suitable candidates for clinical trials in MVID patients. Substances like vincristine (cytostatic drugs) and chlorpromazine (neuroleptic drugs) are already approved drugs for treatment in children, yet not for the use in MVID.